FUJIFILM Toyama Chemical began a phase III clinical trial of Avigan in Japan in March 2020, for COVID-19 patients with non-severe pneumonia. The company conducted randomized, placebo-controlled, single-blind comparative study*1 to evaluate the efficacy and safety of Avigan.
The median value of primary endpoints, using 156 individuals as analysis targets, were 11.9 days for the Avigan group and 14.7 days for the placebo group. FUJIFILM Toyama Chemical confirmed, with a statistically significant difference (p value = 0.0136), that the administration of Avigan to COVID-19 patients with non-serious pneumonia demonstrates shorter time to resolution. The adjusted hazard ratio*2 showed 1.593 (95% confidence interval of 1.024 –2.479). No new safety concerns were noted in this trial.
FUJIFILM Toyama Chemical will conduct a detailed analysis of the data obtained in this trial, and will work to file Application for Partial Changes to include the additional indication in as early as October.
Approved for manufacture and sale in Japan as an influenza antiviral drug, Avigan, selectively inhibits RNA polymerase necessary for influenza virus replication. Due to this mechanism of action, it is expected that Avigan may have an antiviral effect on the new coronavirus, as they are RNA viruses of the same type as influenza viruses.
To meet the requests of the Japanese government to increase stockpiles of Avigan, and by other countries to supply the drug, the Fujifilm Group has been working to increase production of Avigan in collaboration with strategic partners. The Fujifilm Group will work to deliver the treatment drug to COVID-19 patients as soon as possible, and contribute to ending the spread of COVID-19.
*1 A trial in which the subjects are randomly allocated to a group receiving the actual drug or a group receiving a placebo, and a comparative investigation is made without informing the subjects about the drugs that had been allocated to them. In this trial, a comparison was made by administering Avigan and a placebo to the subjects, in addition to standard treatment.
*2 An indicator showing the relative risk of event occurrence by comparison is called a hazard ratio, and a ratio that adjusted the differences in background between the groups compared is called an adjusted hazard ratio. The hazard ratio used in this trial indicates the possibility of symptom relief and virologic responses turning negative. If the hazard ratio exceeds 1, administration of Avigan is judged to be more favorable.
® Tablets>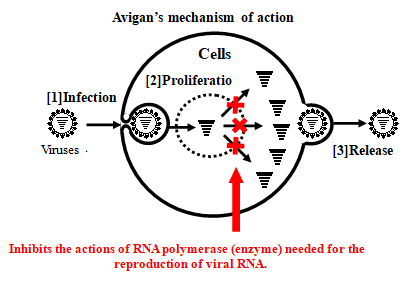
An anti-influenza drug developed by FUJIFILM Toyama Chemical that obtained domestic manufacturing and marketing approval in March 2014 with the treatment of new or reemerging influenza as the indication. The Japanese government has already stockpiled Avigan in preparation for the outbreak of the novel influenza.
For inquiries about this media release, please contact:
<Media>
FUJIFILM Holdings Corporation,
Corporate Communications Division, PR Group
Phone: +81-3-6271-2000
###